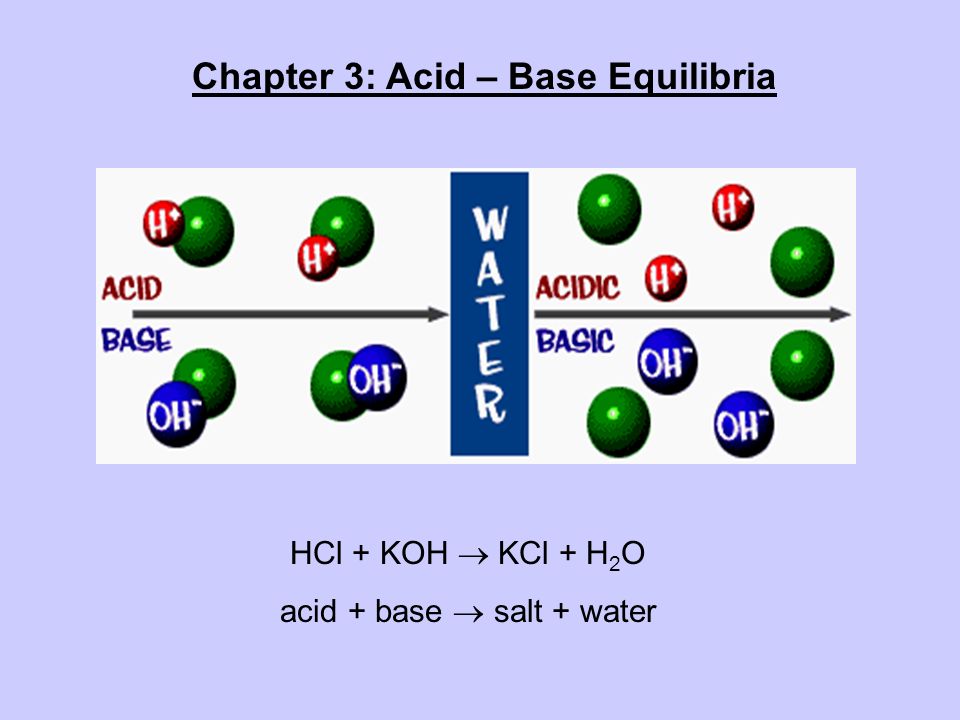
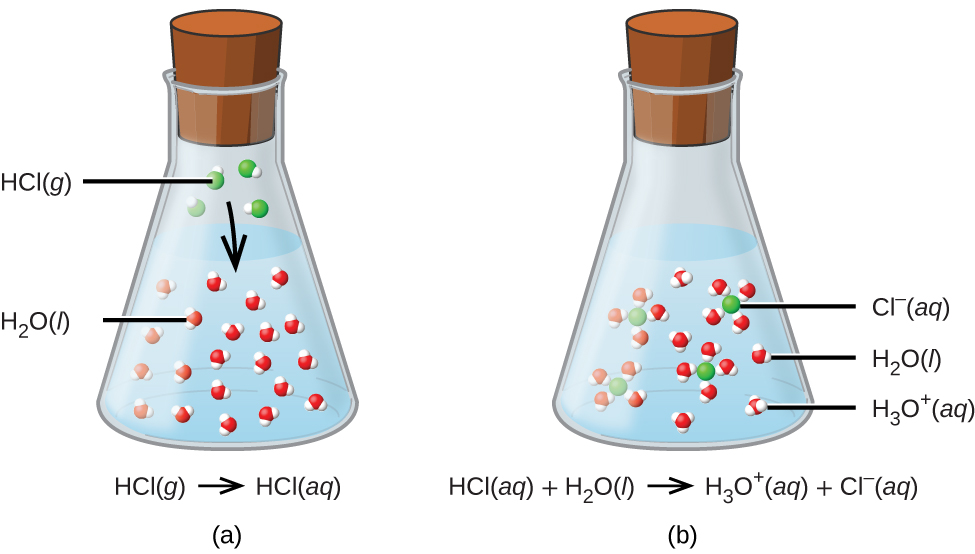
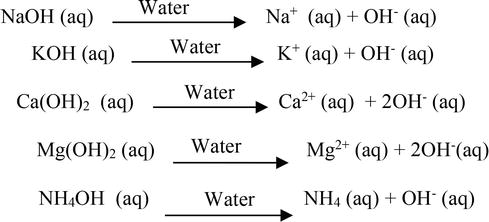
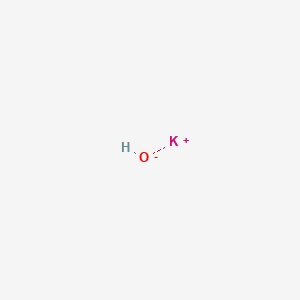Question Video: Calculating the Concentration of Nitric Acid via Titrating against a Known Volume of Potassium Hydroxide | Nagwa

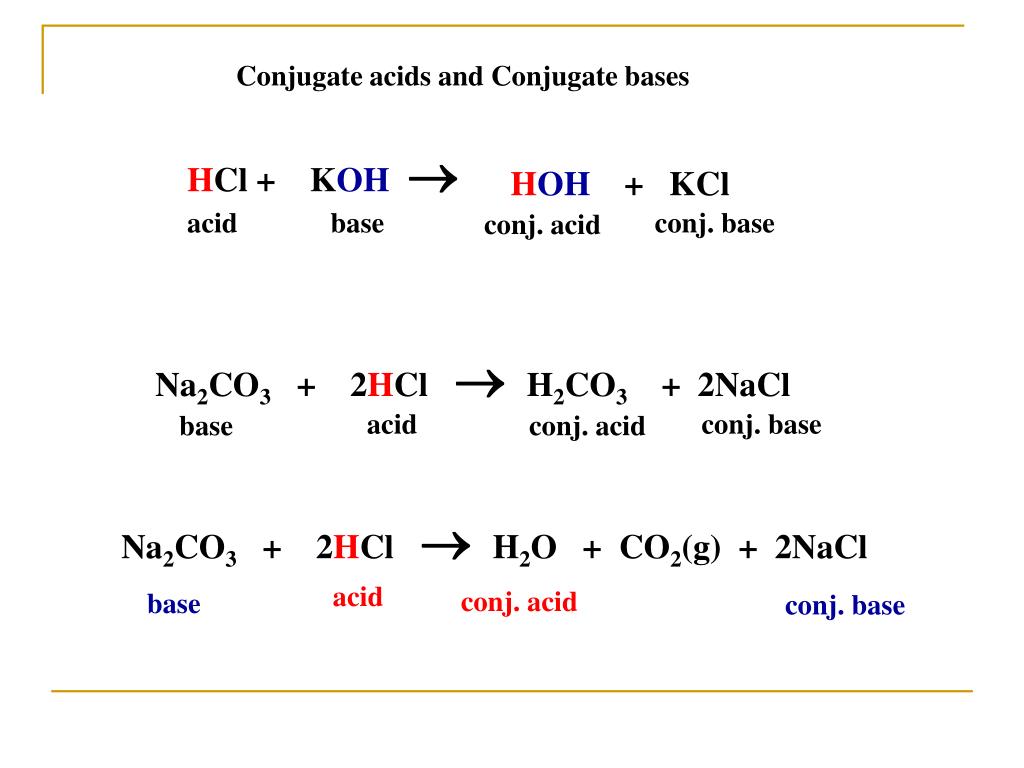
Write the neutralization reaction for the following acid and base: HCl_{(aq)} and KOH_{(aq)}. | Homework.Study.com

Solubility Data for the KOH–K2CO3–K3VO4–H2O System at (313.15 and 353.15) K | Journal of Chemical & Engineering Data