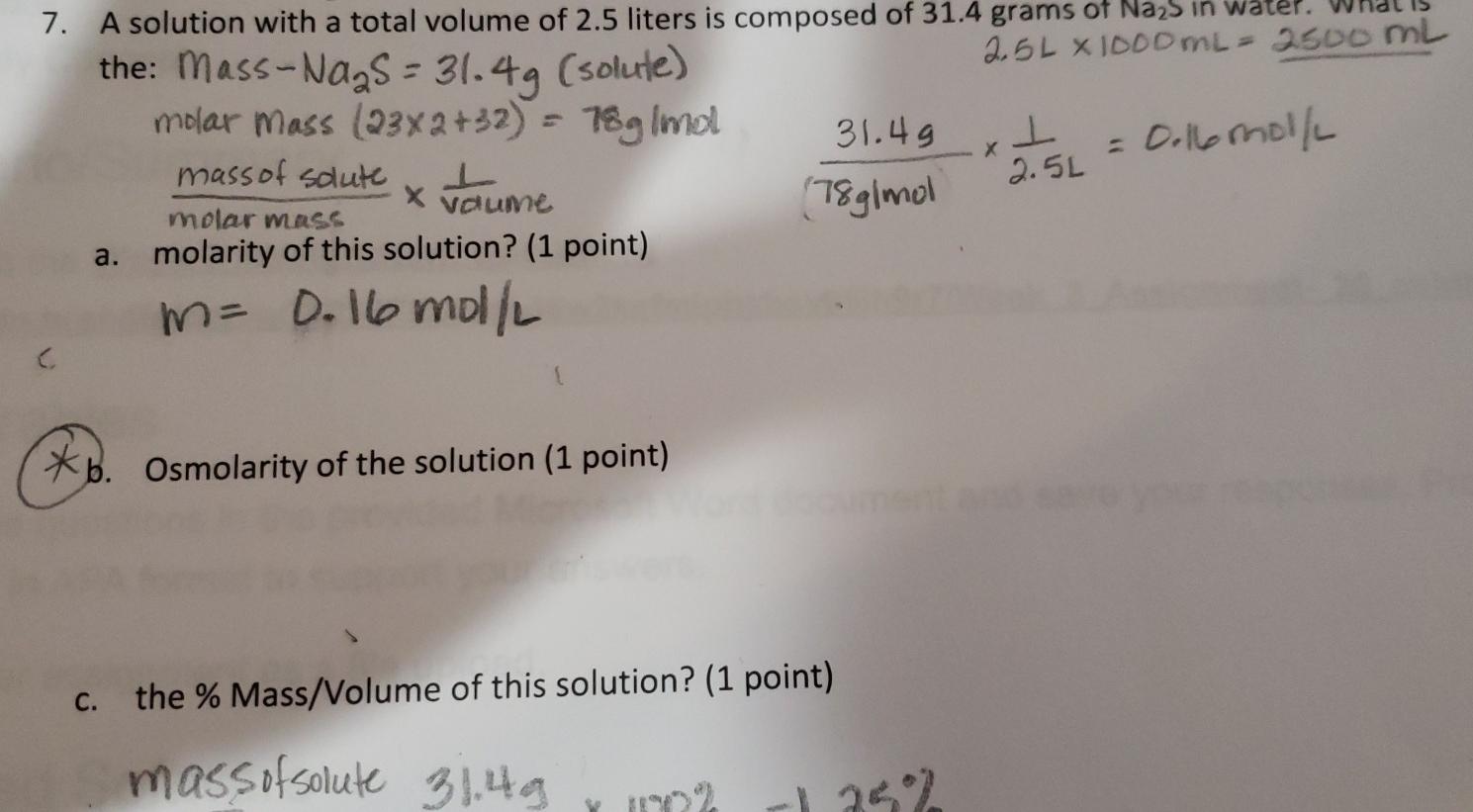Bio-Zone Pond Enzyme Treatment – Ecofriendly Water Cleaner with Natural Enzyme, Fish Waste, Cloudiness –450 Grams Treats 15,000 Liters

SOLVED: points) Initially 15 grams of salt are dissolved into 10 liters of water: Brine with concentration of salt 3 grams per liter is added at a rate of 4 liters per

Molarity Molarity = grams / molar mass / Liters Liters = grams / molar mass/ Molarity Grams = Molarity x Liters x molar mass. - ppt download
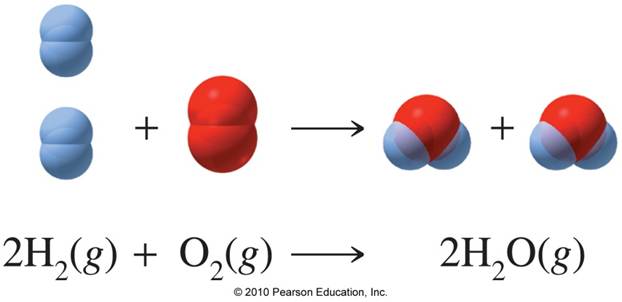
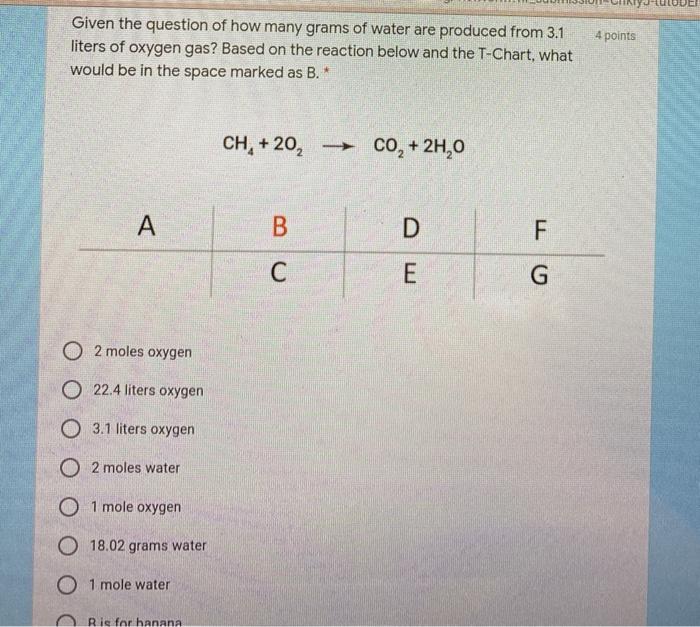
How many liters of water can be made from 55 grams of oxygen gas and an excess of hydrogen at a pressure of 12.4 atm and a temperature of 85C? | Socratic
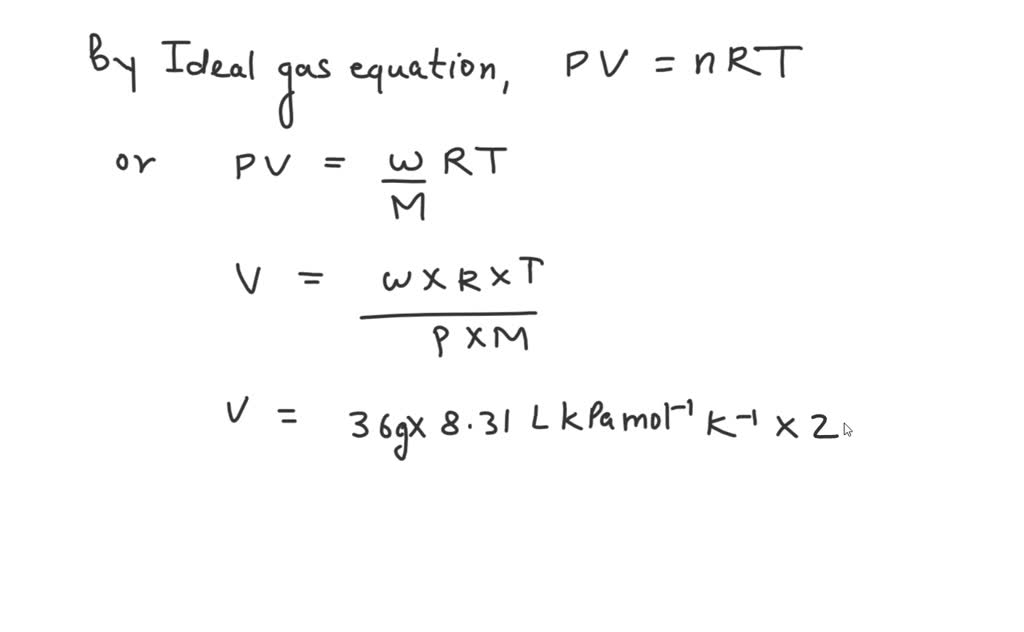
SOLVED: 36 grams of water vapor takes up how many liters at room temperature and pressure (293 K and 100 kPa?)

100% Pure Natural And Mineral Rich Water Bottle For Drinking, 1 Liters Bottle Weight: 10 Grams (G) at Best Price in Asansol | Sanjit Modi & Co.